
Studies from genetically modified animal models corroborate this notion, as Akt1 −/− mice were shown to be defective in exercise-induced cardiac hypertrophy. On the other hand, physiological hypertrophy, such as that induced by exercise, utilizes a different signaling pathway, mainly through phosphoinositide 3-kinase (PI3K), Akt protein kinase B (PKB), and the mammalian target of rapamycin (mTOR). Similarly, activation of the mitogen-activated protein kinases (MAPK) c-Jun N-terminal kinase (JNK) was also shown to contribute to pathological hypertrophic maladaptive gene expression. Once activated, calcineurin tightly binds and dephosphorylates conserved serine residues at the N-terminus of cytoplasmic NFAT transcription factors, permitting their translocation to the nucleus and activation of pathological hypertrophic gene expression. Sustained elevation of calcium ions (Ca +2) downstream of GPCR (αq/α11 subclass) is sensed by calmodulin (Cam) and conveyed to calcineurin, an obligate dimer of regulatory and catalytic subunits with phosphatase activity. A good example supporting this notion is the calcineurin-nuclear factor of activated T cells (NFAT) signaling axis, which was shown to activate pathological hypertrophy. This strongly suggests that a cardiomyocyte undergoing physiological hypertrophy uses different signaling pathways than another one undergoing pathological hypertrophy. On the other hand, G-protein-coupled receptors (GPCR)-activating molecules, such as catecholamines, angiotensin II, and endothellin-1, are linked to the ominous progression to heart failure, and hence have been the target of many pharmacological antagonists. The first are targeted by growth factors, such as insulin-like growth factor-1 (IGF-1) and transforming growth factor beta (TGF-β), which act on tyrosine kinase-coupled receptors (RTKs) and are responsible for the eutrophic, as well as adaptive (physiological), myocyte growth. Based on their target receptor, humoral stimuli can be nested under 2 major groups. Humoral stimuli, on the other hand, act on cell surface receptors, triggering downstream second messenger cascades, finally culminating in cellular hypertrophic response and the associated gene expression program. They do this by binding to the Z-disc myofilament anchor proteins, α-actinin and telethonin, and tethering them to calcineurin, a calcium-dependent phosphatase that was shown to directly induce cardiomyocyte hypertrophy by downstream transcriptional pathways. Calsarcins were shown to couple the cardiac skeletal apparatus to signaling molecules that can directly influence gene expression. A good example of mechanosensitive molecules that have gained attention in recent years are a family of Z-disc-specific proteins called calsarcins, also known as myozenins. The sarcomeric Z-disc and its associated proteins have been suggested to drive mechanical stress-induced signal transduction, a process referred to as “mechanotransduction”. Mechanical stress is thought to induce a hypertrophic response downstream of mechanosensitive molecules. Īt the cellular level, cardiomyocyte hypertrophy is characterized by an increase in cell size, enhanced protein synthesis, and heightened organization of the sarcomere. Ventricular hypertrophy is hence considered as a predictor of cardiovascular morbidity and mortality. Additionally, myocardial supply-demand mismatch secondary to increased myocardial oxygen consumption of the hypertrophic heart further predisposes to multiple cardiovascular ailments, including arrhythmias, myocardial infarction, cerebrovascular events, and sudden death. These are foreboding signs of the development heart failure and pathological remodeling. 
However, in the presence of chronic stressful conditions such as hypertension and valvular disease, a form of pathological hypertrophy develops, which is characterized by excessive increase in ventricular dimensions, accompanied by myocardial dysfunction and fibrosis. 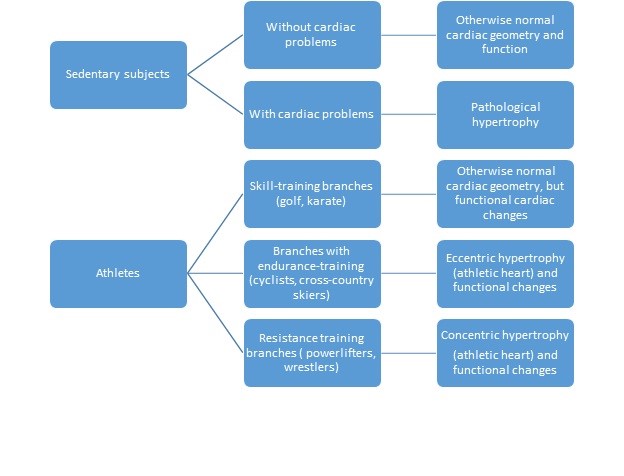
Physiological hypertrophy of the heart can ensue as a result of exercise or pregnancy, and is deemed mild and/or reversible. Hypertrophic growth of the heart is an adaptive response to hemodynamic stress, which is believed to have a compensatory role to enhance cardiac performance and diminish ventricular wall tension and oxygen consumption.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
